| # | Date | Analyst Firm | Upside/Downside | Price Target Change | Rating Change | Current Rating |
|---|
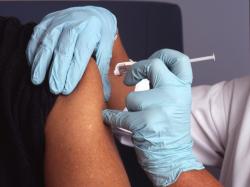
The CDC's Advisory Committee on Immunization Practices recommends Merck's Capvaxive for adults 65+ and certain at-risk ...

, European Commission's Decision Is Expected In Q3

- Bloomberg

- Reuters

The FDA issued a Complete Response Letter for Daiichi Sankyo and Merck's BLA for patritumab deruxtecan targeting EGFR-mutat...

The U.S. Food and Drug Administration (FDA) has issued a Complete Response Letter (CRL) for the Biologics License Application (...

On Tuesday, Merck Group revealed an unexpected decision to end a Phase III trial of its cancer drug Xevinapant. This move threa...

Innovative technology represents a breakthrough scientific achievement for highly contagious respiratory disease